Potency of Leaf Water Extract Karamunting (Rhodomyrtus tomentosa) for Enhancing GLUT4 Translocation in Diabetic Rats
Abstract
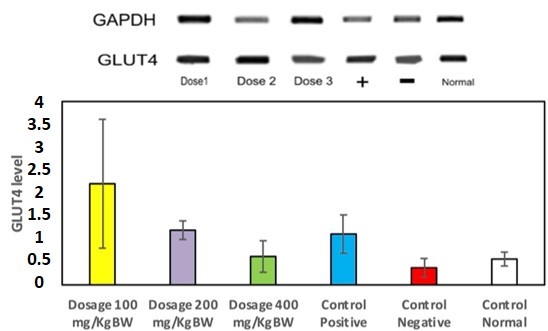
Insulin resistance and damage in the pancreas are associated with type 2 diabetes, which is a chronic disease affecting health, with 422 million people affected in 2016 and 1.5 million deaths related to diabetes in 2012. Several proinflammatory mediators, such as TNF-a andIL-6, may be involved in insulin resistance in T2DM. GLUT4 is one of the glucose transporters that help promote glucose uptake. GLUT4 activation is insulin-dependent in regulating glucose uptake. Karamunting (Rhodomyrtus tomentosa (Ait.) Hassk.) leaf water extract consists of alkaloids, flavonoids, triterpenoids, saponins, and tannins. This study proposes to investigate the potential of karamunting (R. tomentosa) leaf water extract to enhance glucose uptake by modulating GLUT4 translocation in the serum and skeletal muscle of diabetic rats. The dose variations used in this study were 100 mg/kgBW, 200 mg/kgBW, and 400 mg/kgBW. Based on the results, oral administration of karamunting water extract increases GLUT4 expression in serum. It increases GLUT4 mRNAlevels in the skeletal muscle of the diabetic rat. It decreases TNF-a and IL-6 expression, with a 100 mg/kg BW dosage being the most effective. Meanwhile, 100 mg/KgBW is the most effective dosage for decreasing HOMA IR. These results indicate that karamunting (R. tomentosa) leaf water is antidiabetic by lowering TNF-a, IL-6, and HOMA IR in diabetic rats while increasing GLUT4.
References
Brus, M., R. Frangež, M. Gorenjak, P. Kotnik, Ž. Knez, and D. Škorjanc (2021). Effect of Hydrolyzable Tannins on Glucose-Transporter Expression and Their Bioavailability in Pig Small-Intestinal 3D Cell Model. Molecules, 26(2); 345
Bryant, N. J., R. Govers, and D. E. James (2002). Regulated Transport of the Glucose Transporter GLUT4. Nature Reviews Molecular Cell Biology, 3(4); 267–277
Endarini, L. H. (2016). FarmakognisidanFitokimiaKomprehensif. Pusdik SDM Kesehatan (in Indonesia), Jakarta
Fatkhurrohman, I., A. M. Aman, H. Umar, A. Bukhari, H. N. HS, H. Sanusi, others, and A. A. Zainuddin (2025). The Role of Glycemic Control and Vitamin D3 Levels in Modulating mRNA Expression of GLUT4 and IL-6 in Newly Diagnosed Type 2 Diabetes Mellitus Patients: Diabetes Mellitus. Journal of Neonatal Surgery, 14(32S)
Febriyanto, G. and M. I. Saleh (2021). Efektivitas Antidiabetes Fraksi Air Daun Karamunting (Rhodomyrtus tomentosa (Ait.) Hassk.) terhadap Kadar Glukosa Darah dan Sekresi Insulin pada Tikus Model Diabetes. Jurnal Ilmiah Kedokteran Wijaya Kusuma, 10(1); 57–70 (in Indonesia)
Feriotto, G., F. Tagliati, V. Costa, M. Monesi, C. Tabolacci, S. Beninati, and C. Mischiati (2024). ????-Pinene, a Main Component of Pinus Essential Oils, Enhances the Expression of Insulin-Sensitive Glucose Transporter Type 4 in Murine Skeletal Muscle Cells. International Journal of Molecular Sciences, 25(2); 1252
Furuya, D. T., A. C. Poletto, R. R. Favaro, J. O. Martins, T. M. T. Zorn, and U. F. Machado (2010). Anti inflammatory Effect of Atorvastatin Ameliorates Insulin Resistance in Monosodium Glutamate–treated Obese Mice. Metabolism, 59(3); 395–399
Garvey, W. T., L. Maianu, J. H. Zhu, G. Brechtel-Hook, P. Wallace, and A. D. Baron (1998). Evidence for Defects in the Trafficking and Translocation of GLUT4 Glucose Transporters in Skeletal Muscle as a Cause of Human Insulin Resistance. The Journal of Clinical Investigation, 101(11); 2377–2386
Ghasemzadeh, A., H. Z. E. Jaafar, M. F. M. Bukhori, M. H. Rahmat, and A. Rahmat (2018). Assessment and comparison of phytochemical constituents and biological activities of bitter bean (Parkia speciosa Hassk.) collected from different locations in Malaysia. Chemistry Central Journal, 12(1); 12
Hasibuan, R., S. Ilyas, and S. Hannum (2015). Effect of Leaf Extract Haramonting (Rhodomyrtus tomentosa) to Lower Blood Sugar Levels in Mice Induced by Alloxan. 8(6); 284–291
He, M., J. Jiang, S. Liu, and H. Cheng (2013). Effect of Iron Supplementation on Glucose Transporter 4 Expression in Adipose Tissue and Skeletal Muscle of Pregnant Rats. Open Journal of Obstetrics and Gynecology, 3(6); 500–507
He, Z. X., Z. W. Zhou, Y. Yang, T. Yang, S. Y. Pan, J. X. Qiu, and S. F. Zhou (2015). Overview of Clinically Approved Oral Antidiabetic Agents for the Treatment of Type 2 Diabetes Mellitus. Clinical and Experimental Pharmacology and Physiology, 42(2); 125–138
Hotamisligil, G. S., P. Arner, J. F. Caro, R. L. Atkinson, and B. M. Spiegelman (1995). Increased Adipose Tissue Expression of Tumor Necrosis Factor-???? in Human Obesity and Insulin Resistance. The Journal of Clinical Investigation, 95(5); 2409–2415
Huang, S. and M. P. Czech (2007). The GLUT4 Glucose Transporter. Cell Metabolism, 5(4); 237–252
Iwata, M., T. Haruta, I. Usui, Y. Takata, A. Takano, T. Uno, and M. Kobayashi (2001). Pioglitazone Ameliorates Tumor Necrosis Factor-????–Induced Insulin Resistance by a Mechanism Independent of Adipogenic Activity of Peroxisome Proliferator–Activated Receptor-????. Diabetes, 50(5); 1083–1092
Kouidhi, S., R. Berrhouma, K. Rouissi, S. Jarboui, M.-S. Clerget-Froidevaux, I. Seugnet, others, and A. B. Elgaaied (2013). Human Subcutaneous Adipose Tissue Glut4 mRNA Expression in Obesity and Type 2 Diabetes. Acta Diabetologica, 50(2); 227–232
Kurauti, M. A., J. M. Costa-Júnior, S. M. Ferreira, G. J. Santos, C. H. G. Sponton, E. M. Carneiro, and A. C. Boschero (2017). Interleukin-6 Increases the Expression and Activity of Insulin-Degrading Enzyme. Scientific Reports, 7(1); 46750
Leahy, J. L. (2005). Pathogenesis of Type 2 Diabetes Mellitus. Archives of Medical Research, 36(3); 197–209
Leguisamo, N. M., A. M. Lehnen, U. F. Machado, M. M. Okamoto, M. M. Markoski, G. H. Pinto, and B. D. Schaan (2012). GLUT4 Content Decreases Along with Insulin Resistance and High Levels of Inflammatory Markers in Rats with Metabolic Syndrome. Cardiovascular Diabetology, 11(1); 100
Lv, Y., W. Ren, Y. Zhang, Y. Huang, J. Hao, K. Ma, Y. Ma, and X. Yang (2020). Antidiabetic Effects of a Lipophilic Extract Obtained from Flowers of Wisteria sinensis by Activating Akt/GLUT4 and Akt/GSK3????. Food & Nutrition Research, 64; 10–29219
Moller, D. E. (2000). Potential Role of TNF-???? in the Pathogenesis of Insulin Resistance and Type 2 Diabetes. Trends in Endocrinology & Metabolism, 11(6); 212–217
Muoio, D. M. and C. B. Newgard (2008). Molecular and Metabolic Mechanisms of Insulin Resistance and ????-Cell Failure in Type 2 Diabetes. Nature Reviews Molecular Cell Biology, 9(3); 193–205
Na-Phatthalung, P., M. Teles, S. P. Voravuthikunchai, L. Tort, and C. Fierro-Castro (2018). Immunomodulatory Effects of Rhodomyrtus tomentosa Leaf Extract and Its Derivative Compound, Rhodomyrtone, on Head Kidney Macrophages of Rainbow Trout (Oncorhynchus mykiss). Fish Physiology and Biochemistry, 44(2); 543–555
NHS (2022). High Blood Sugar (Hyperglycaemia). Retrieved August 24, 2022
Nikzamir, A., A. Palangi, A. Kheirollaha, H. Tabar, A. Malakaskar, H. Shahbazian, and M. Fathi (2014). Expression of Glucose Transporter 4 (GLUT4) Is Increased by Cinnamaldehyde in C2c12 Mouse Muscle Cells. Iranian Red Crescent Medical Journal, 16(2); e13426
Pal, M., M. A. Febbraio, and M. Whitham (2014). From Cytokine to Myokine: The Emerging Role of Interleukin-6 in Metabolic Regulation. Immunology & Cell Biology, 92(4); 331–339
Peng, Q., G. Liu, P. Li, X. Wu, Q. Zeng, and C. Zhu (2020). A Potential Role for GLUT4 in Predicting Sepsis in Critically Ill Children. Research Square; 1–20. Preprint, Version 1
Rehman, K., M. S. H. Akash, A. Liaqat, S. Kamal, M. I. Qadir, and A. Rasul (2017). Role of Interleukin-6 in Development of Insulin Resistance and Type 2 Diabetes Mellitus. Critical Reviews in Eukaryotic Gene Expression, 27(3); 229–236
Roglic, G. and W. H. Organization (2016). Global Report on Diabetes. World Health Organization
Sari, D., M. Romi, N. Arfian, J. Yunus, W. Setyaningsih, and R. Yuniartha (2021). Hands on The LAB HOTLAB From Animal to Data. Fakultas Kedokteran Universitas Gadjah Mada, Yogyakarta
Senn, J. J., P. J. Klover, I. A. Nowak, and R. A. Mooney (2002). Interleukin-6 Induces Cellular Insulin Resistance in Hepatocytes. Diabetes, 51(12); 3391–3399
Sinaga, E., S. E. Rahayu, Suprihatin, and Yenisbar (2019). Potensi Medisinal Karamunting (Rhodomyrtus tomentosa). UNAS Press, Jakarta
Tilg, H. and A. R. Moschen (2008). Inflammatory Mechanisms in the Regulation of Insulin Resistance. Molecular Medicine, 14(3); 222–231
Vo, T. S. and D. H. Ngo (2019). The Health Beneficial Properties of Rhodomyrtus tomentosa As Potential Functional Food. Biomolecules, 9(2); 76
Wang, T., J. Wang, X. Hu, X. Huang, and G.-X. Chen (2020). Current Understanding of Glucose Transporter 4 Expression and Functional Mechanisms. World Journal of Biological Chemistry, 11(3); 76–98
Wei, F., W. Zhang, S. Kang, P. Liu, Y. Yao, W. Liu, and X. Yang (2024). Phenolic Constituents with Glucose Uptake and GLUT4 Translocation Bioactivities from the Fruits of Cordia dichotoma. Journal of Agricultural and Food Chemistry, 72(29); 16298–16311
Winarsih, S., A. Z. T. Munaf, A. D. Treasa, D. Noviani, D. Y. N. Hidayati, M. Nooryanto, and S. Sutrisno (2023). Effects of Ethanolic Extract of Rhodomyrtus tomentosa Leaves on Cytokines Levels in Puerperal Infection Mice Induced with Escherichia coli. In AIP Conference Proceedings, volume 2634. page 020066
Yagasaki, K. (2014). Anti-diabetic Phytochemicals That Promote GLUT4 Translocation via AMPK Signaling in Muscle Cells. Nutrition and Aging, 2(1); 35–44
Zhou, M., K. William H., H. Canhua, P. Yinbo, S. Jie, and X. Wang (2023). Bioactivity and Mechanisms of Flavonoids in Decreasing Insulin Resistance. Journal of Enzyme Inhibition and Medicinal Chemistry, 38(1); 2199168
Authors

This work is licensed under a Creative Commons Attribution 4.0 International License.