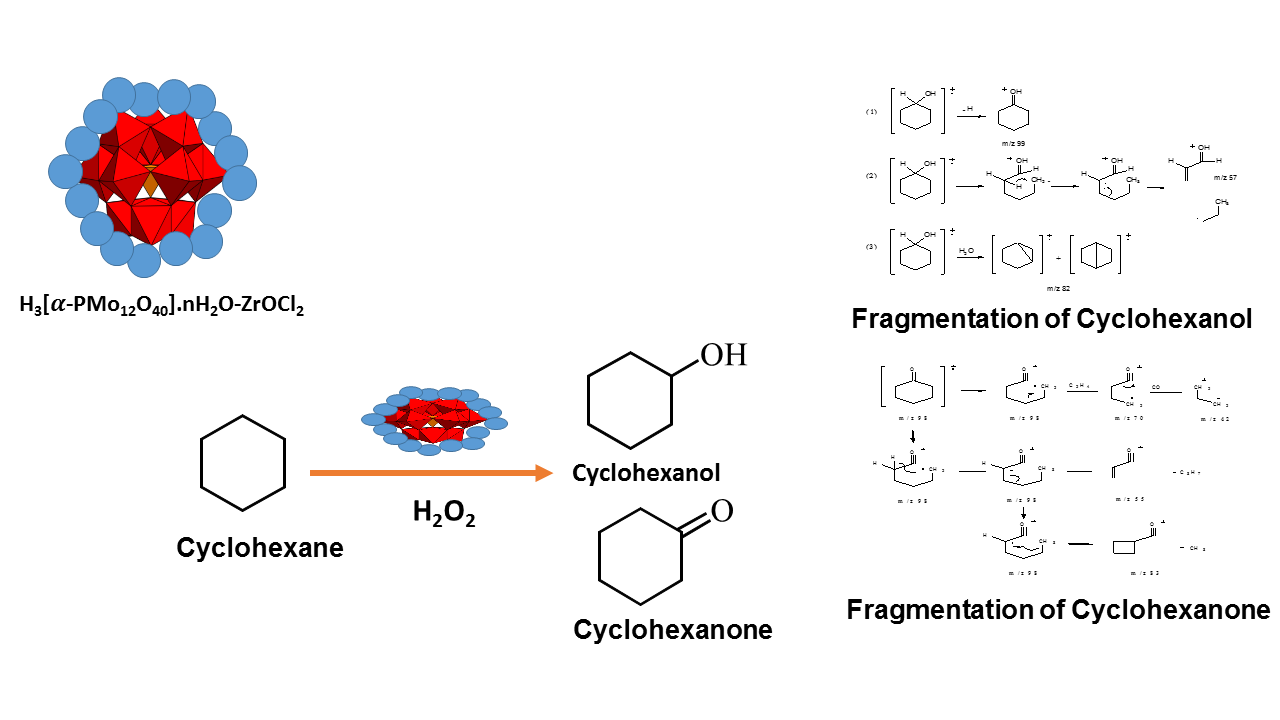
Conversion of cyclohexane to cyclohexanol and cyclohexanone using H3[PMo12O40].nH2O-ZrOCl2 as catalyst
Abstract
Synthesis and preparation polyoxometalate compound H3[-PMo12O40].nH2O supported with ZrOCl2 at various weights of ZrOCl2 i.e. 0.25 g, 0.50 g, 0.75 g, 1.00 g, 1.25 g, 0.01 g and 0.05 g to form H3[-PMo12O40].nH2O/ZrOCl2 have been conducted. These compound than was characterized through functional group analysis using FT-IR spectrophotometer and XRD analysis. The results showed that the optimal preparation was H3[-PMo12O40].nH2O/ZrOCl2 with support 0.05 g. FT-IR spectrum of H3[-PMo12O40].nH2O/ZrOCl2 show wavenumber at 1033.85 cm-1 for vibration P-O, 887.26 cm-1 for vibration M=O, 840.96 cm-1 and 655.80 cm-1 for vibration Mo-O-Mo. The existence of support was identified at wavenumber 1404.18 cm-1for vibration Zr-OH and 478.35 for vibration Zr-O-Zr. XRD powder analysis showed that material H3[-PMo12O40].nH2O/ZrOCl2 was amorphous material. Material H3[-PMo12O40].nH2O/ZrOCl2 was applied in oxidation of cyclohexane using hydrogen peroxide as oxidant. Oxidation process was optimized through reaction time H2O2 amount, temperature and catalyst weight. The results showed that the highest conversion in the using of H3[-PMo12O40].nH2O/ZrOCl2 catalyst found at 2 hours reaction time, 3 mL H2O2, 80oC temperature and 0.038 g catalyst with conversion 99.18 %. Selectivity of best reaction was 6.96 % for cyclohexanol and 24.9% for cyclohexanone, which was identified by GC-MS.
Keywords : H3[-PMo12O40].nH2O-ZrO2,cyclohexane, cyclohexanol, cyclohexanone.